In the field of chemical engineering, heat transfer operation is a fundamental concept that plays a crucial role in a wide range of processes. Both heat and temperature are crucial ideas in every branch of science as well as in students’ everyday lives. Humans interact with heat and temperature daily, but many have serious misconceptions about them. From distillation columns to heat exchangers, understanding the principles of heat transfer is essential for designing, optimizing, and operating these systems efficiently.
In this blog post, we will provide an introduction to heat transfer operation, covering the basic concepts and terminology that you need to know. Whether you’re a student of chemical engineering or a professional in the field, this post will serve as a useful resource to refresh your knowledge of heat transfer concepts. So, let’s dive in and explore the world of heat transfer in chemical engineering!
Table of Contents
What is heat transfer?
Heat transfer is the process of exchanging thermal energy between two or more objects. This is an important process in many industries, including manufacturing, energy production, and transportation. Heat transfer operations are a crucial part of many engineering processes, as they determine how heat moves through a system and how efficient that system is at transferring heat.
In simple words, Heat Transfer is
Energy in transit due to temperature difference.
What are the types of heat transfer?
There are three main types of heat transfer: conduction, convection, and radiation.
Conduction occurs when heat is transferred through a material without any movement of the material itself. This process is common in solids, as the molecules in a solid are closely packed together and can transfer energy quickly from one to the other.
Convection occurs when heat is transferred through a fluid, such as air or water, by the movement of that fluid. This process is often seen in heating and cooling systems, as air or water is heated or cooled and then circulated through a building or system.
Radiation occurs when heat is transferred through electromagnetic waves, such as infrared radiation. This process is responsible for the heat we feel from the sun and heat lamps and is also used in many industrial processes such as drying, curing, and sterilization.
To understand the concept of conduction, convection, and radiation better, let’s look at the following figure:

What is the Conduction?
Conduction is a type of heat transfer that occurs when heat is transferred from one object to another through direct contact. This process happens when a molecule in a material collides with another molecule, transferring heat energy from the hotter molecule to the cooler one. The rate of heat transfer through conduction depends on several factors, including the thermal conductivity of the material, the surface area of contact, and the temperature difference between the two objects. To understand the concept of conduction better, let’s look at the following figure:
In this figure, we have two blocks of different materials in contact with each other. The block on the left is made of metal, which has a high thermal conductivity, while the block on the right is made of wood, which has a lower thermal conductivity. The metal block is at a higher temperature than the wood block, so heat flows from the metal block to the wood block through direct contact. As a result, the metal block loses some of its thermal energy, and the wood block gains thermal energy.
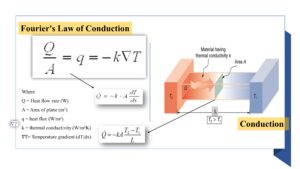
Fourier’s Law
The rate of heat transfer through conduction can be calculated using Fourier’s Law, which states that
“The rate of heat transfer is proportional to the temperature gradient across the material and the thermal conductivity of the material.”
Simply,
“The heat flux is proportional to the temperature gradient.”
See the formula for Faurier’s law in the above figure. In summary, conduction is a type of heat transfer that occurs through direct contact between objects. It is an important process in many engineering applications and can be controlled by adjusting the materials used and the surface area of contact between objects.
What is the Convection?
Convection is a type of heat transfer that occurs in fluids, such as liquids and gases. It is the movement of hot fluid (or gas) from one place to another, carrying heat energy with it. The movement is typically caused by differences in temperature or density within the fluid. Convection can occur in two ways: natural convection and forced convection.
Natural Convection
Natural convection occurs when the movement of fluid is driven by buoyancy forces caused by temperature differences. For example, when a pot of water is heated on a stove, the water near the bottom of the pot gets hot and expands, becoming less dense than the cooler water above it. This causes the hot water to rise to the top of the pot and the cooler water to sink to the bottom, creating a convection current.
Forced Convection
Forced convection occurs when the movement of fluid is driven by an external force, such as a fan or pump. For example, when air is blown over a hot object, such as a computer processor, the heat from the object is transferred to the air. The movement of the air is then forced by a fan, carrying the heat energy away from the object and dissipating it into the surrounding environment.
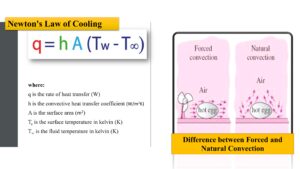
The rate of heat transfer by convection can be calculated using Newton’s law of cooling.
Newton’s law of cooling
The rate of heat loss in a body is directly proportional to the temperature differential between the body and its surroundings.
Key points
-
This formula is known as Newton’s law of cooling, and it applies to both natural and forced convection.
-
The convective heat transfer coefficient, h, is a measure of the ability of the fluid to transfer heat by convection, and it depends on properties such as the fluid velocity, temperature, and viscosity, as well as the geometry of the object and the surface roughness.
-
The convection heat transfer coefficient h is not a property of the fluid.
-
Convection is an important process in many engineering applications, such as in cooling systems for engines and electronic devices, as well as in weather patterns and ocean currents. Understanding how convection works can help engineers design more efficient and effective systems, while also improving our understanding of natural phenomena.
What is the Radiation?
Radiation is a type of heat transfer that occurs through electromagnetic waves, which can travel through a vacuum or any transparent medium, such as air or glass. Unlike conduction and convection, which require a physical medium, radiation can occur in a vacuum, such as in space. All objects with a temperature greater than absolute zero (-273.15°C or 0 Kelvin) emit thermal radiation in the form of electromagnetic waves. The amount and wavelength of radiation emitted depend on the temperature and emissivity of the object.
Emissivity is a measure of how well an object emits radiation, with a value of 1 representing a perfect emitter and 0 representing a perfect reflector.
In addition to emitting radiation, objects can also absorb radiation from their surroundings. The amount of radiation absorbed depends on the wavelength of the radiation and the absorptivity of the object.
Absorptivity is a measure of how well an object absorbs radiation, with a value of 1 representing a perfect absorber and 0 representing a perfect transmitter.
Stefan-Boltzmann law:
The Stefan-Boltzmann law of radiation governs the rate of heat transfer by radiation emitted. The Stefan-Boltzmann law is an important equation in radiation heat transfer that describes the relationship between the temperature of an object and the rate at which it emits thermal radiation. It is given by the equation:

Key Points
-
The Stefan-Boltzmann law shows that the rate of radiant heat transfer increases with the fourth power of the absolute temperature of the object.
-
This means that small changes in temperature can lead to significant changes in the rate of radiant heat transfer.
-
The law also shows that the rate of radiant heat transfer is proportional to the surface area of the object and the emissivity, which describes how well the object emits radiation.
-
The Stefan-Boltzmann law is used in many engineering applications, such as in the design of thermal insulation, solar collectors, and radiative cooling systems.
-
It is also used in astrophysics to study the properties of stars and other celestial objects.
Radiation is an important process in many engineering applications, such as in the design of thermal insulation and solar energy systems. Understanding how radiation works can help engineers design more efficient and effective systems, while also improving our understanding of natural phenomena, such as the Earth’s energy balance and the behavior of stars.
Important formulas in heat transfer
There are several important formulas in heat transfer that are used to calculate various parameters related to the transfer of heat. Here are some of the most used formulas:
Fourier’s Law: This formula describes the rate of heat conduction through a material. It can be written as:
q = -kA(dT/dx)
Where q is the heat flux, k is the thermal conductivity of the material, A is the cross-sectional area of the material, and dT/dx is the temperature gradient.
Newton’s Law of Cooling: This formula describes the rate of heat transfer between a solid surface and a fluid. It can be written as:
q = hA(Ts – Tf)
Where q is the heat transfer rate, h is the heat transfer coefficient, A is the surface area, Ts is the temperature of the solid surface, and Tf is the temperature of the fluid.
Heat Transfer Coefficient: This formula describes the amount of heat transferred per unit of time and per unit area of a surface. It can be written as:
h = q/(A(T_s – T_f))
Where h is the heat transfer coefficient, q is the heat transfer rate, A is the surface area, T_s is the temperature of the solid surface, and T_f is the temperature of the fluid.
Biot Number: This formula describes the ratio of internal resistance to external resistance in a heat transfer process. It can be written as:
Bi = hL/k
Where Bi is the Biot number, h is the heat transfer coefficient, L is a characteristic length scale, and k is the thermal conductivity.
Nusselt Number: This formula describes the mass transfer coefficient for convection. It can be written as:
Nu = hL/k
Where Nu is the Nusselt number, h is the heat transfer coefficient, L is a characteristic length scale, and k is the thermal conductivity.
Reynolds Number: This formula describes the ratio of inertial forces to viscous forces in a fluid. It can be written as:
Re = ρVL/μ
Where Re is the Reynolds number, ρ is the density of the fluid, V is the velocity of the fluid, L is a characteristic length scale, and μ is the dynamic viscosity.
These formulas are essential for understanding and designing heat transfer operations in various industries, such as chemical engineering, mechanical engineering, and materials science.
Application of Heat transfer operation
Heat transfer operations are widely used in many industries and applications. Heat transfer operations are used in many different industries and applications. In manufacturing, heat transfer is used to melt and shape materials, such as in the production of glass or metal. In energy production, heat transfer is used to convert fuel into energy, such as in the burning of coal or the fission of nuclear fuel. In transportation, heat transfer is used to cool engines and exhaust systems, as well as to provide heat to the cabin of a vehicle. Here are a few examples:
HVAC (Heating, Ventilation, and Air Conditioning) Systems: Heat transfer operations are used in HVAC systems to maintain comfortable indoor temperatures and air quality. These systems use convection and sometimes radiation to transfer heat between the air and other surfaces in a building. For example, radiators or baseboard heaters transfer heat by convection to warm up a room.
Power Generation: Heat transfer operations are used in power generation to convert thermal energy into electricity. In thermal power plants, steam is produced by heating water using heat from burning fossil fuels or nuclear reactions. The steam is then used to power turbines that generate electricity. This process involves conduction, convection, and radiation.
Chemical Processing: Heat transfer operations are used in chemical processing to control the temperature of reactions and to facilitate heat exchange between fluids. For example, heat exchangers are used to transfer heat between two fluids, such as in cooling or heating a chemical process.
Food Processing: Heat transfer operations are used in food processing to preserve food, improve its quality, and enhance its flavor. For example, pasteurization uses heat transfer to kill harmful bacteria in food and extend its shelf life.
Automotive Industry: Heat transfer operations are used in automotive engines to maintain the optimum operating temperature of the engine components. Radiators and cooling systems transfer heat away from the engine to prevent overheating.
Electronics Industry: Heat transfer operations are used in the electronics industry to cool electronic components and prevent them from overheating. Heat sinks, fans, and other cooling systems are used to transfer heat away from the components and maintain their operating temperatures.
These are just a few examples of the many applications of heat transfer operations in various industries. Heat transfer operations play a crucial role in the efficient and safe functioning of many processes and systems.
Conclusion
In conclusion, heat transfer operations are a vital aspect of many industries and engineering processes. Understanding the different types of heat transfer, as well as their applications and limitations, is crucial for ensuring the success and efficiency of these processes. By optimizing heat transfer, engineers can improve the performance, safety, and cost-effectiveness of their systems and ultimately contribute to the advancement of technology and industry. Efficient heat transfer is critical to the success of many engineering processes, as it can affect the performance, safety, and cost-effectiveness of a system. Engineers must carefully consider the type of heat transfer required for a given application, as well as the materials and design of the system to achieve the desired outcome.
———————-****************———————
Important questions and answer
Here are 25 important questions and answers related to heat transfer operations in chemical engineering, that might be useful for competitive exams (GATE) and interviews:
Question 1: What is heat transfer?
Answer: Heat transfer is the process of exchanging thermal energy between two or more objects or systems.
Question 2: What are the three modes of heat transfer?
Answer: The three modes of heat transfer are conduction, convection, and radiation.
Question 3: What is conduction heat transfer?
Answer: Conduction heat transfer is the transfer of thermal energy through a material without any net motion of the material itself.
Question 4: What is Fourier’s law of heat conduction?
Answer: Fourier’s law of heat conduction states that the rate of heat transfer through a material is proportional to the temperature gradient and the cross-sectional area of the material.
Question 5: What is thermal conductivity?
Answer: Thermal conductivity is the measure of a material’s ability to conduct heat.
Question 6: What is convection heat transfer?
Answer: Convection heat transfer is the transfer of thermal energy due to the motion of fluids.
Question 7: What is natural convection?
Answer: Natural convection is the transfer of thermal energy due to the motion of fluids caused by buoyancy effects.
Question 8: What is forced convection?
Answer: Forced convection is the transfer of thermal energy due to the motion of fluids caused by an external source, such as a fan or pump.
Question 9: What is the Nusselt number?
Answer: A Nusselt number is a dimensionless number that relates the convective heat transfer coefficient to the thermal conductivity of a material.
Question 10: What is radiation heat transfer?
Answer: Radiation heat transfer is the transfer of thermal energy through electromagnetic waves.
Question 11: What is the Stefan-Boltzmann law?
Answer: The Stefan-Boltzmann law states that the rate of radiation heat transfer from a surface is proportional to the fourth power of its absolute temperature.
Question 12: What is emissivity?
Answer: Emissivity is a measure of how well a material emits thermal radiation.
Question 13: What is absorption?
Answer: Absorption is the process by which a material absorbs radiation.
Question 14: What is reflection?
Answer: Reflection is the process by which a material reflects radiation.
Question 15: What is the transmission?
Answer: Transmission is the process by which a material allows radiation to pass through it.
Question 16: What is thermal resistance?
Answer: Thermal resistance is a measure of how difficult it is for thermal energy to flow through a material.
Question 17: What is the heat transfer coefficient?
Answer: The heat transfer coefficient is a measure of the rate of heat transfer between a fluid and a solid surface.
Question 18: What is a heat exchanger?
Answer: A heat exchanger is a device that transfers thermal energy between two fluids that are at different temperatures.
Question 19: What are the types of heat exchangers?
Answer: The types of heat exchangers are shell and tube, plate, and finned tube.
Question 20: What is a counterflow heat exchanger?
Answer: A counterflow heat exchanger is a type of heat exchanger in which the two fluids flow in opposite directions.
Question 21: What is a parallel flow heat exchanger?
Answer: A parallel flow heat exchanger is a type of heat exchanger in which the two fluids flow in the same direction.
Question 22: What is a cross-flow heat exchanger?
Answer: A cross-flow heat exchanger is a type of heat exchanger in which the two fluids flow perpendicular to each other.
Question 23: What is the effectiveness of a heat exchanger?
Answer: The effectiveness of a heat exchanger is the ratio of the actual heat transfer to the maximum possible heat transfer.
Question 24: What is the log mean temperature difference (LMTD) method?
Answer: The LMTD method is a common method used to calculate the heat transfer rate in a heat.
Question 25: What is the difference between natural and forced convection?
Answer: Natural convection occurs when the motion of the fluid is caused by buoyancy forces due to temperature differences. Forced convection occurs when an external force, such as a pump or a fan, is used to move the fluid.
Question 26: What is the unit of thermal conductivity?
Answer: The unit of thermal conductivity is W/mK.
Question 27: What is Fourier’s law of heat conduction?
Answer: Fourier’s law of heat conduction states that the rate of heat transfer through a material is proportional to the temperature gradient.
Question 28: What is the unit of heat transfer coefficient?
Answer: The unit of heat transfer coefficient is W/m2K.
Question 29: What is Nusselt number?
Answer: Nusselt number is a dimensionless parameter that describes the convective heat transfer coefficient of a fluid.
Question 30: What is the difference between thermal conductivity and thermal diffusivity?
Answer: Thermal conductivity is a measure of a material’s ability to conduct heat, while thermal diffusivity is a measure of how quickly a material can respond to changes in temperature.
Question 31: What is Reynolds’s number?
Answer: Reynolds number is a dimensionless parameter that describes the flow regime of a fluid.
Question 32: What is Prandtl number?
Answer: Prandtl number is a dimensionless parameter that describes the ratio of momentum diffusivity to thermal diffusivity in a fluid.
Question 33: What is Biot number?
Answer: Biot number is a dimensionless parameter that describes the ratio of internal thermal resistance to external thermal resistance in a solid.
Question 34: What is Graetz number?
Answer: Graetz number is a dimensionless parameter that describes the ratio of convective heat transfer to conductive heat transfer in a fluid flowing through a pipe.
Question 35: What is the difference between adiabatic and isothermal processes?
Answer: Adiabatic processes occur without the transfer of heat, while isothermal processes occur at a constant temperature.
Question 36: What is the difference between a steady-state and a transient heat transfer process?
Answer: A steady-state heat transfer process occurs when the temperature of a system does not change over time, while a transient heat transfer process occurs when the temperature of a system changes over time.
Question 37: What is the difference between a single-pass and a multi-pass heat exchanger?
Answer: In a single-pass heat exchanger, the fluid flows through the heat exchanger only once. In a multi-pass heat exchanger, the fluid flows through the heat exchanger multiple times.
Question 38: What is a counter-current heat exchanger?
Answer: A counter-current heat exchanger is a type of heat exchanger in which the hot and cold fluids flow in opposite directions.
Question 39: What is a parallel-flow heat exchanger?
Answer: A parallel-flow heat exchanger is a type of heat exchanger in which the hot and cold fluids flow in the same direction.
Question 40: What is the effectiveness of a heat exchanger?
Answer: The effectiveness of a heat exchanger is the ratio of the actual heat transfer to the maximum possible heat transfer.
Question 41: What is fouling in heat exchangers?
Answer: Fouling is the accumulation of unwanted material, such as dirt, scale, or biological growth, on the surface of a heat exchanger, which can reduce heat transfer efficiency.
Question 42: What is the difference between a finned tube heat exchanger and a plain tube heat exchanger?
Answer: A finned tube heat exchanger has fins on the outside of the tubes, which increases the surface area and improves
Question 43: Why is heat transfer important in chemical engineering?
Answer: Heat transfer systems for chemical processes must be created to maximize process efficiency. A failure to concentrate on efficiency can result in unnecessarily higher costs because the heat transfer step in many chemical processes consumes a lot of energy.
Question 44: What are the 3 types of heat transfer?
Answer: Three methods exist for transferring heat: conduction, convection, and radiation.
Question 45: What are heat transfer introductions and basic concepts?
Answer: Radiation, convection, and conduction are the three mechanisms by which heat can be transferred. Every time there is a temperature difference between states of matter, heat transfer takes place. Heat transfer only happens in the direction of decreasing temperature, or from a hot object to a cold item.
——-******——-
Must Read:


5 thoughts on “Introduction to heat transfer operation || Basic Concepts and Applications || Chemical Engineering || 2023”